Structural analysis of impurities in pharmaceuticals

We perform the structural analysis of impurities in drug substances and drug products, based on ICH-Q3A and ICH-Q3B guidelines.
Features
- We study the structural analyses of impurities according to the requests from our clients.
We present reliable data to our clients, in accordance with the japan's Reliability Criteria *1 and GMP (Japan, USA, and Europe).
*1: Article 43 of the Regulation for Enforcement of the Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices - For a compound with the molecular weight of approximately 500, its structural analysis can be performed using a sample amount of 0.5 mg.
Please do not hesitate to ask us, even if peaks are very small and poorly separated each other in your chromatography. - We propose the structures of impurities, estimated by using mass-spectrometer*2 with high-resolution, high-scan speed, and multistage MS.
*2: Thermo Fisher Scientific, Orbitrap Eclipse Tribrid Mass Spectrometer with Orbitrap, quadrupole, and ion-trap, with maximal 500,000 (FWHM) resolution at m/z 200.
Our Experience
Structural analysis
- Related substances and impurities in drug substances and drug products
- Forced degradation products of drug substances
- Degradation products generated during stability studies
- Unknown compounds derived from manufacturing problems
- Extractables and leachables derived from container closure system
Analysis of foreign matters contaminated in drug substances and drug products
Case Study
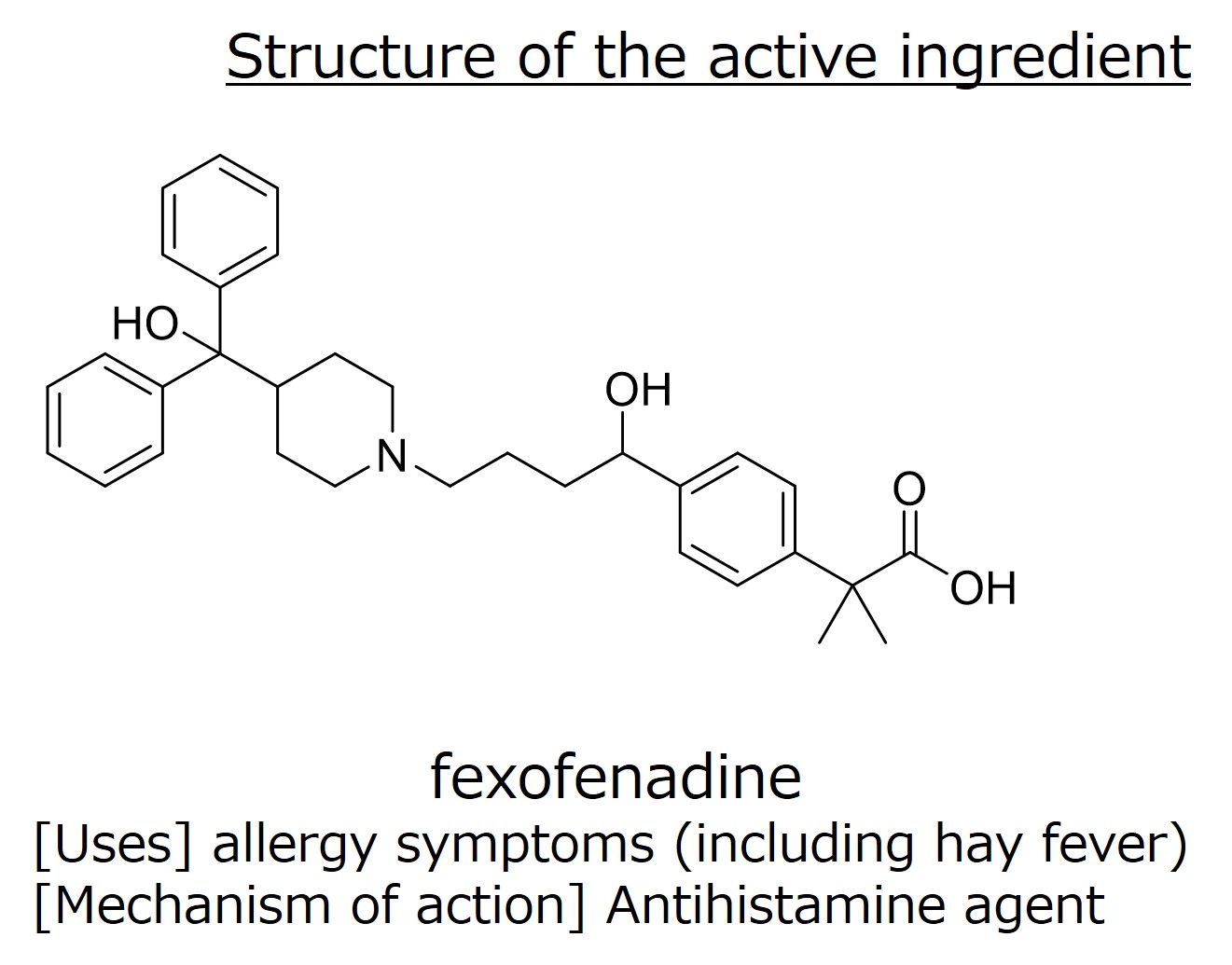
An enforced oxidation product (at 60°C for 10 hours with 30% hydrogen peroxide solution) of a pharmaceutical for hay fever (active ingredient: Fexofenadine) was used. As shown in Fig. 1, several degradation products (Peaks 1-6 and Peak 8) were detected as well as the main component Fexofenadine (Peak 7).
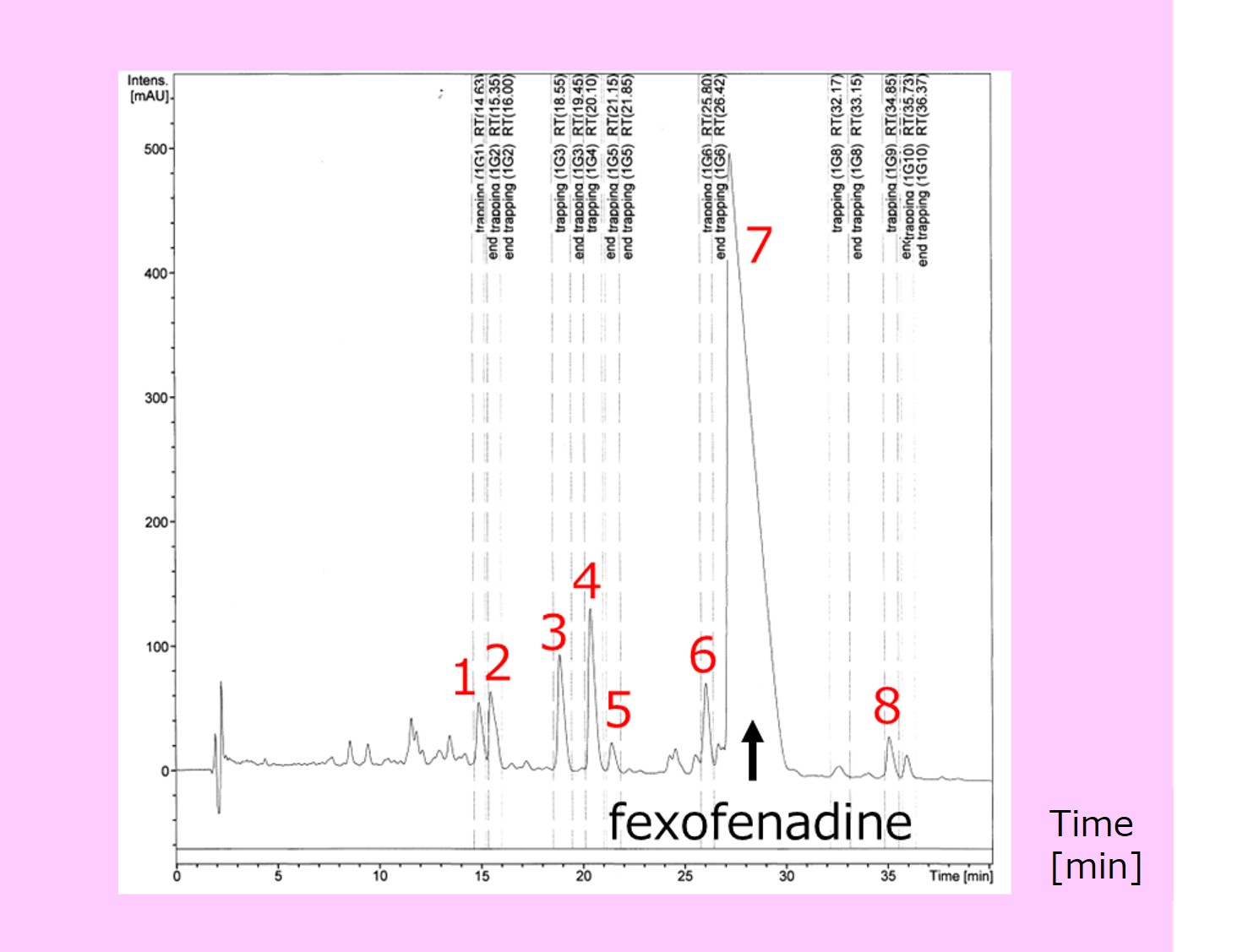
We determined the composition formula by precision mass spectrometry.
Among them, we show the structural analysis of Peak 3. First, we found that Peak 3 has one more oxygen (O) than fexofenadine.
| Precision mass | Composition formula | |
|---|---|---|
| fexofenadine | 502.2948 | C32H40NO4 |
| Peak 3 | 518.2898 | C32H40NO5 |
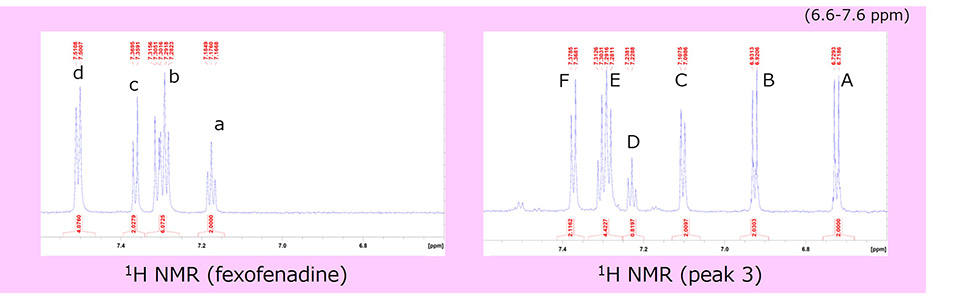
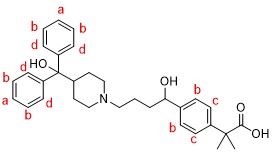
Next, we separated Peak 3 from other peaks and acquired its 1H NMR spectrum. It was significantly different from that of fexofenadine in the aromatic region (6.6~7.7ppm), suggesting that the aromatic proton of fexofenadine was oxidized. Since the chemical shift values, number of protons, and coupling constants were analyzed, the strucure of Peak 3 was identified as the Structural formula 2, below.
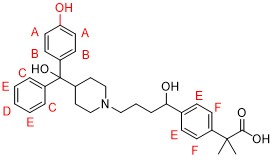
In addition, we can perform more detailed structural analyses using a combination of precision mass spectrometry and various types of two-dimensional NMR (COSY, HSQC, HMBC etc.).
We can also perform the analyses of structure elucidation, intermolecular interactions and conformation of medium-sized molecules with a cyclic structure, using NMR measurement.
Instrument


Contact Us for Services
