Biopharmaceutical Characterization, Quality Testing, and Stability Testing : Antibodies, ADCs (Antibody-Drug Conjugates), Bispecific Antibodies, and so on

We have a proven track record for evaluating diverse biopharmaceuticals, including monoclonal antibodies, ADCs (Antibody-Drug Conjugates), and bispecific antibodies. We also have experience on-site technical training at Japanese and global client’s sites.
Features
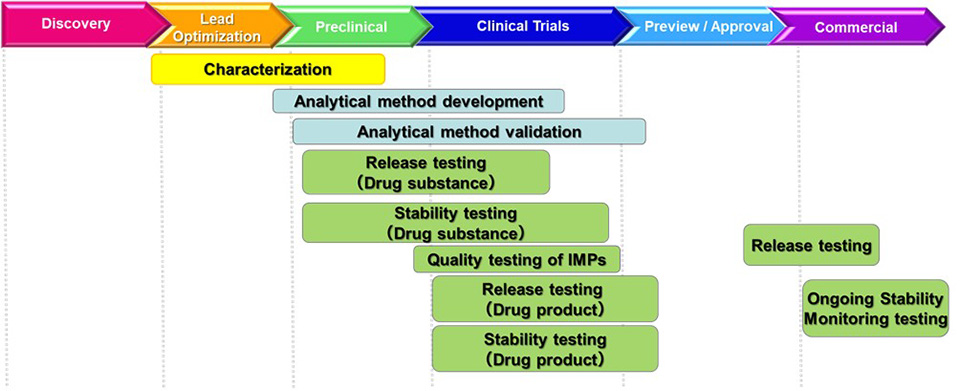
We provide analytical support from early research and development stage of biopharmaceuticals to their commercial stage according to the requests from our clients. We have extensive experience in characterization, quality testing and stability testing under GMP and the Japan's Reliability Criteria *1.
Characterization
- Characterization of the target molecules using high‑resolution LC‑MS for amino acid sequencing, glycan analysis, and structural elucidation of impurities
Quality Testing
- For drug substances and drug products, analytical method development and validation studies, as well as release testing for clinical trial and commercial purposes
- Biological activity testing using cell‑based bioassays and surface plasmon resonance analysis (SPR)
Stability Testing
- Long‑term testing, accelerated testing, stress testing, and photostability testing are conducted in accordance with ICH Q1A/Q1B and ICH Q5C applied for biotechnologicals.
- Temperature and humidity in the stability storage facilities are monitored by centralized NASTER System.
Sample Handling
- Powder containment facilities (EC2) enable our handling of highly potent compounds up to OEL Category 5.
- BSL-2 laboratories enable our handling of biohazard samples.
Analysis items
Characterization
| Test items | Methods & Instruments | |
|---|---|---|
| Structure analysis | Amino acid composition | Amino acid analyzer |
| N‑terminal and C‑terminal sequencing | LC‑MS, LC‑MS/MS | |
| Free thiols and disulfide bonds | LC‑MS, LC‑MS/MS | |
| Drug‑to‑antibody ratio (DAR) profile | HPLC, UPLC, LC‑MS, LC‑MS/MS | |
| Glycan composition, glycan structural analysis, glycosylation sites, glycan mapping | HPLC, UPLC, LC‑MS, LC‑MS/MS | |
| Physicochemical properties | Size variant and Molecular weight | SDS‑PAGE, SE‑HPLC, CE‑SDS, intact mass by LC‑MS |
| Charge variants | cIEF, icIEF | |
| Chromatographic profile | HPLC, UPLC | |
| Immunological properties (Immunological binding) | ELISA | |
| Activity | Potency | ELISA, Cell-based assay, SPR |
| Cytotoxic activity (ADCC, CDC) | Cell-based assay | |
| Impurities | Product‑related impurities (fragments, aggregates, deamidation and oxidation) | LC‑MS, LC‑MS/MS |
| Process‑related impurities (host cell proteins, residual DNA, etc.) | HPLC, UPLC, LC‑MS, LC‑CAD, ELISA, real‑time PCR | |
Quality testing
| Test items | Methods & Instruments | |
|---|---|---|
| Identity | Primary structure (Amino acid analysis, Peptide mapping) | HPLC, UPLC, LC‑MS |
| Glycan patterns and composition | HPLC, UPLC, LC‑MS | |
| Molecular weight | SDS‑PAGE, SE‑HPLC, CE‑SDS, intact mass by LC‑MS | |
| Electrophoretic profile | SDS‑PAGE, cIEF, CE‑SDS | |
| N‑terminal and C‑terminal analyses | LC‑MS, LC‑MS/MS | |
| Bioassay | Cell-based assay | |
| Immunoassay | ELISA | |
| Purity | Size variants | SE‑HPLC, SDS‑PAGE, CE‑SDS, RP‑HPLC |
| Charge variants | IEX‑HPLC, IEF, cIEF, icIEF | |
| Host cell proteins, residual DNA | ELISA, Real-time PCR | |
| Process‑related impurities | HPLC, UPLC, LC‑MS, LC‑CAD | |
| Potency | Bioactivity | ELISA, Cell-based bioassay, SPR |
| Content | Protein quantification | Spectrophotometry, Variable pathlength UV‑Vis (SoloVPE), BCA assay (USP‑compliant) |
| Others | Aggregate assessment | Dynamic light scattering (Zetasizer) |
| Drug‑to‑antibody ratio (DAR) | HIC-UV, LC-MS | |
| Microbiological test | Sterility, Bacterial endotoxins, Microbial limits, Mycoplasma testing (JP/USP/EP) | |
Instruments

MS: Orbitrap Eclipse、Thermo Fisher Scientific






Relevant guidelines
- *1: Article 43 of the Regulation for Enforcement of the Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices
Biopharmaceuticals-related
-
QUALITY OF BIOTECHNOLOGICAL PRODUCTS:STABILITY TESTING OF BIOTECHNOLOGICAL/BIOLOGICAL PRODUCTS (Recommended for Adoption at Step 4 of the ICH Process on 30 November 1995 by the ICH Steering Committee)
-
SPECIFICATIONS : TEST PROCEDURES AND ACCEPTANCE CRITERIA FOR BIOTECHNOLOGICAL/BIOLOGICAL PRODUCTS (Recommended for Adoption at Step 4 of the ICH Process on 10 March 1999 by the ICH Steering Committee)
-
DERIVATION AND CHARACTERISATION OF CELL SUBSTRATES USED FOR PRODUCTION OF BIOTECHNOLOGICAL/BIOLOGICAL PRODUCTS (Recommended for Adoption at Step 4 of the ICH Process on 16 July 1997 by the ICH Steering Committee)
Stability test
-
STABILITY TESTING OF NEW DRUG SUBSTANCES AND PRODUCTS Q1A(R2) (Current Step 4 version dated 6 February 2003)
-
STABILITY TESTING: PHOTOSTABILITY TESTING OF NEW DRUG SUBSTANCES AND PRODUCTS (Recommended for Adoption at Step 4 of the ICH Process on 6 November 1996 by the ICH Steering Committee)
Analysis method validation
-
VALIDATION OF ANALYTICAL PROCEDURES Q2(R2) (Final Version Adopted on 1 November 2023)
-
VALIDATION OF ANALYTICAL PROCEDURES: TEXT AND METHODOLOGY Q2(R1) (Current Step 4 version Parent Guideline dated 27 October 1994 (Complementary Guideline on Methodology dated 6 November 1996 incorporated in November 2005))
Contact Us for Services
