Exosome Quality Evaluation Services

Extracellular vesicles (EVs), including exosomes, are increasingly attracting attention for their therapeutic use and functionality as drug delivery system (DDS). Several EV-based products are under development, and exosome therapies are already being implemented in some advanced clinics and medical institutions. Beyond pharmaceutical and healthcare industries, EVs are widely being used in cosmetics, food and other sectors. Thus, we provide comprehensive quality evaluation services on exosome/EV products for medical use; development of pharmaceuticals; and applications in cosmetics and food. We can provide reliable and trustworthy data on EV quality evaluation.
Key Features
- Quality testing is conducted in accordance with ISO 9001 (Quality management systems—Requirements) or GMP, as applicable.
- We provide end‑to‑end support from analytical method development to validation and to testing.
- We preform product-specific evaluation testing according to the requests from our clients.
Test Menu
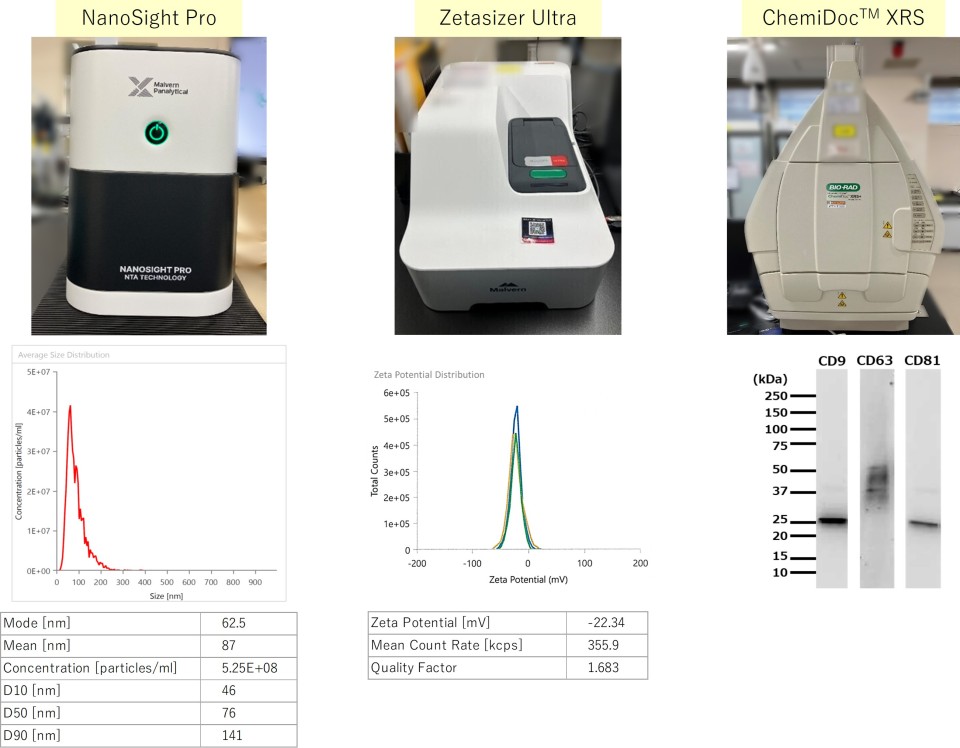
| Test Items | Analytical Techniques |
|---|---|
| Content (particle count) | Nanoparticle tracking analysis (NTA) |
| Particle size distribution | Nanoparticle tracking analysis (NTA); Dynamic light scattering (DLS) |
| Surface markers | Western blotting |
| Surface charge (zeta potential) | Electrophoretic light scattering |
| Encapsulated miRNA | PCR; ddPCR |
| Encapsulated proteins | Western blotting |
| Potency assays | ELISA; PCR; cell‑based assays |
| Impurities | ELISA; PCR; LC/MS, etc. |
| Total protein | BCA assay |
| Sterility test* | Direct inoculation; membrane filtration |
| Rapid sterility test* | Microcolony method |
| Mycoplasma test* | Method A; Method C |
| Endotoxins test* | Gel‑clot; turbidimetric; chromogenic techniques |
- *compliant with GMP
Instruments and representative data
Points to consider in providing samples derived human cell cultures
When sending samples obtained from the human body to us, please review and get approval from your ethical review committee, in accordance with the "Ethical Guidelines for Medical and Biological Research Involving Human Subjects", and take necessary measures required by this guideline.
Contact Us for Services
