Quality evaluation of regenerative medicine

We provide support to our clients with a variety of analytical techniques from early research and development stage of regenerative medicine.
Sumika Chemical Analysis Service (SCAS) provides a wide range of quality evaluation services for regenerative medicine.
Features
Quality evaluation and release testing of new drugs required for approval applications are conducted in compliance with pharmaceutical regulations, such as GMP and the Japan’s Reliability Criteria *1.
- *1: Article 43 of the Regulation for Enforcement of the Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices
We can perform analytical method development and method validation, and evaluate the quality of final products as well as raw material cells.
Product-specific evaluation testing will also be conducted according to the requests from our clients.
Our services in the development of regenerative medicine
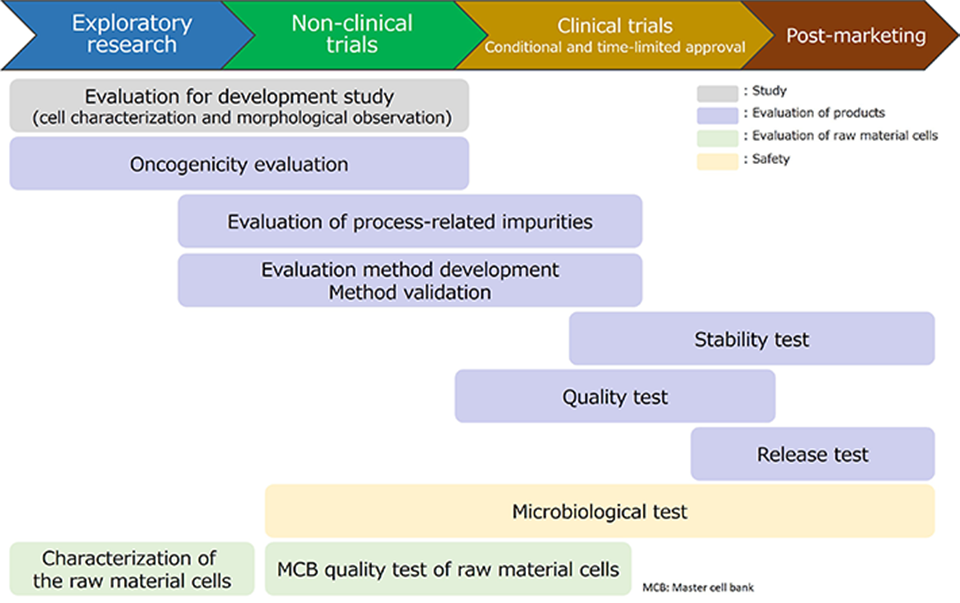
Test items
| Evaluation items | Evaluation items | Analytical technique |
|---|---|---|
| Content | Cell count, cell viability | Cytometry (Hemocytometer/Automated cytometry instrument), Flow cytometry |
| Identity | Appearance, Cell phenotype, Differentiation, Cell type, Agglutination, etc. | Visual observations, Cellular immunostaining, quantitative PCR (qPCR), Droplet Digital PCR (ddPCR), Flow cytometry, Particle size distribution |
| Purity | Cell phenotype, Abnormal growth, etc. | Observations, Immunostaining, qPCR, ddPCR, Flow cytometry |
| Process-related impurities | Process-related impurities (Serum albumin, Antibiotics, Raw material-derived vectors, etc.) | LC/MS, GC/MS, ICP-MS, LC-CAD, ELISA, qPCR, ddPCR |
| Impurities with undesirable physiological activities | Physiologically active substance, etc. | LC/MS, GC/MS, ELISA, Membrane array, etc. |
| Safety | Mycoplasma, Endotoxin, Sterility, etc. | Microbiological tests, Rapid sterility test |
| Virus | qPCR | |
| Residual undifferentiated cells | qPCR, Flow cytometry | |
| Chromosomal aberrations | PCR | |
| Karyotype Analysis (G-band) | ||
| Potency Efficacy |
Protein expression, Secretion of physiologically active substances, Differentiation, Cell phenotype, Cell proliferation, etc. | ELISA, Cell assay, Cellular immunostaining, Visual observations, etc., Flow cytometry |
| Characterization | Localization and differentiation studies of cells | Immunostaining (fluorescent microscopy) using organization sections. |
| Study of cell differentiation and evaluation of storage stability | Live-cell image analysis |
Quality evaluation of gene therapy products
Instruments

(KEYENCE BZ-X700)

(BD FACSLyricTM)

(Bio-Rad QX200)
Our Experiences
Our experiences of testing from FY2014
- Number of Clients: 50 or more
- Number of studies: 1200 or more, including 300 or more feasibility studies
Precautions regarding test samples
Please provide us with the following information in advance.
If samples should be handled under Biosafety level (BSL) 3 or higher or if the safety of samples cannot be ensured, we may not accept them.
- Information on infectivity of samples and pathogenic micro-organisms (origin, BSL, the methods that show non-pathogenicity, status of inactivation, etc.)
- Whether β-lactam antibiotics are used or not
- Whether it is a genetically modified organism or not
Points to consider in providing samples derived from human cultured cells
When sending samples obtained from the human body to us, please review and get approval from your ethical review committee, in accordance with the "Ethical Guidelines for Medical and Biological Research Involving Human Subjects", and take necessary measures required by this guideline.
Contact Us for Services
