Bioanalysis for Oligonucleotides Using Low-Adsorption LC–MS/MS

Sumika Chemical Analysis Service, Ltd. (SCAS) addresses the analytical challenges associated with oligonucleotides and matrix effects by using our optimal sample-preparation techniques. We develop analytical methods to ensure rapid and reliable quantification of oligonucleotides, using advanced systems composed of low-adsorption HPLC and high-sensitivity MS/MS.
Background
Oligonucleotides represent a promising next-generation therapeutic modality, particularly for diseases that have remained difficult to treat with conventional small molecules and biologics.
Quantitative analysis of drug concentrations in biological matrices, which often contain various endogenous contaminants, requires highly sensitive and selective LC-MS/MS methodologies. Using such methoslogies, simultaneous determination of the target nucleic acids and their related metabolites can be achieved. In addition, oligonucleotides have unique structural and physicochemical properties such as non-specific adsorption to analytical devices, making their analyses more difficult.
Features
- For oligonucleotides, which are often challenging to quantify with high sensitivity due to their adsorption to analytical devices, we optimize sample pre-treatment and LC conditions including the selection of ion-pairing reagents, in order to minimize their carryover during analysis and achieve highly accurate and precise assays.
- All analyses are conducted in compliance with regulatory requirements including GLP and the ICH M10 guideline (International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use—M10: Bioanalytical Method Validation and Study Sample Analysis).
- For research and development of oligonucleotides, we offer comprehensive services including cytotoxicity screening during lead discovery, anti-drug antibody (ADA) analysis, cytokine profiling and specification and quality testing of our clients’ target compounds.
Case Studies
The ICH M10 guideline requires both bioanalytical method validation and study sample analysis. Below is an example of our approach to meeting this guideline:
Model Compound
- Fomivirsen
oAntisense nucleic acid drug
oPhosphorothioate (PS) modification
oMW: 6682.4
oSequence: 5'-GCGTTTGCTCTTCTTCTTGCG-3' (21 bases)
Assay Requirements and Suitability
- Quantitation range: 2.00–1000 ng/mL
- Linearity: Compliant
- Accuracy: Compliant
- Selectivity: Compliant
- Carryover: Compliant
Instrumentation
- HPLC: Nexera™ XS inert (Shimadzu Corp.)
- MS: Triple Quad™ 7500 (AB Sciex Pte. Ltd.)
Chromatograms
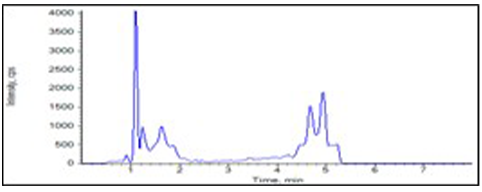
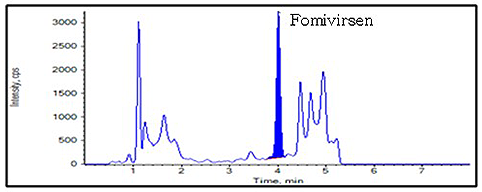
Accuracy and Precision
| Nominal Conc.(ng/mL) | 2 | 5 | 300 | 800 |
|---|---|---|---|---|
| Determined Conc.(ng/mL) | 2.1 | 4.62 | 324 | 832 |
| 1.83 | 4.77 | 315 | 845 | |
| 1.94 | 4.25 | 304 | 803 | |
| Mean S.D. Precision (%) Accuracy (%) |
1.96 | 4.55 | 314 | 827 |
| 0.14 | 0.27 | 10 | 22 | |
| 6.9 | 5.9 | 3.2 | 2.6 | |
| 97.8 | 90.9 | 104.8 | 103.3 |
Related Analytical Services for Oligonucleotides
We also provide a wide range of analyses essential for the development of oligonucleotides:
- ADA (Anti-Drug Antibody) Analysis: ELISA (Enzyme-Linked Immuno-Sorbent Assay), ECL (Electrochemiluminescence), and SPR (Surface Plasmon Resonance) methods.
- Cytokine Analysis: ELISA (including Ella™ automated systems), ECL methods.
- Characterization Tests: Identity, assay, related substances, purity, and biological activity.
- Toxicity Screening: Cytotoxicity, and upon request, phototoxicity and in vitro micronucleus assays.
References
- GLP (MHW Ordinance No. 21): "Ministerial Ordinance on Criteria for Conducting Nonclinical Studies on Drug Safety"
- GCP (MHW Ordinance No. 28): "Ministerial Ordinance on Criteria for the Conduct of Clinical Trials on Drugs"
- Pharmaceuticals and Medical Devices Agency: "ICH M10 Bioanalytical Method Validation and Study Sample Analysis," <000272440.pdf> (accessed 2025.11.28)
Contact Us for Services
