Bioanalysis of Antibody-Drug Conjugates (ADCs)

To understand the pharmacokinetics of antibody-drug conjugates (ADCs), which have complex molecular structures, it is essential to accurately quantify the concentrations of the following components in biological matrices:
- Total antibody
- Conjugated antibody (ADC)
- Unconjugated (free) payload (drug) and its active metabolites
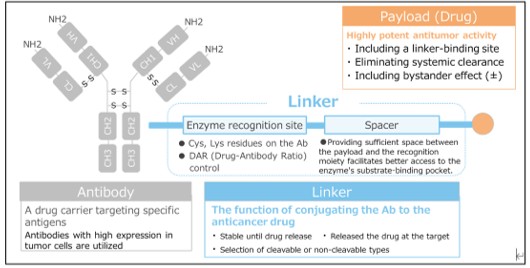
Analytical Strategies for ADC Characterization
Our bioanalytical platform enables the quantification of total antibody, conjugated antibody (ADC), payload in both conjugated and unconjugated forms, and its active metabolites. The quantification of conjugated payload is achieved using a technology that combines immunoprecipitation (IP) and liquid chromatography-tandem mass spectrometry (LC-MS/MS) as well as conventional ligand-binding assays (LBAs) and other LC-MS/MS methods.
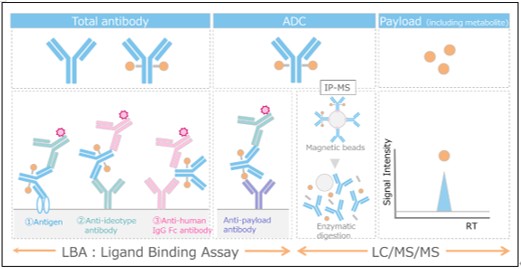
Features
Leveraging our extensive expertise in both small- and large-molecule bioanalysis, we support our clients’ ADC development from drug discovery through to regulatory approval, as follows:
- Proposal of analysis strategies tailored to your research objectives at each R&D stage
- Quantification of trace components utilizing high-sensitivity MSs (TQ7500, SCIEX)
- Precise LBAs with our experienced and robust procedures
- Rapid analyses with automated platforms that consist of the high-speed immunoassay system Gyrolab™ (Gyros Protein Technologies) and automated IP system KingFisher Apex System (Thermo Fisher Scientific Inc.).
- Analyses in compliance with GLP and the ICH M10 guideline on bioanalytical method validation and samples analysis.
Overview of Pharmacokinetic Studies in ADC R&D (SCAS services)
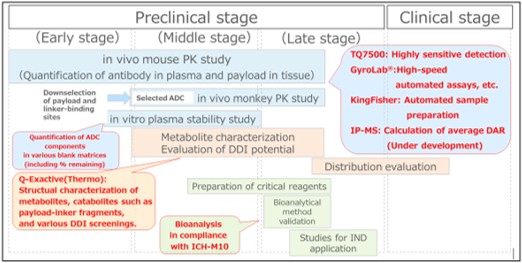
Our Experience
- We have extensive experience in quantification of conjugated payloads by using IP-LC-MS/MS and of unconjugated payloads using LC-MS/MS.
- In regulatory studies for analyzing clinical samples, we have extensive experience in bioanalytical method validation and sample analysis. This includes quantification of ADCs, total antibodies and ADAs using LBAs, as well as quantification of payloads using LC-MS/MS.
Additional Analytical Services
We provide a wide range of related analytical services essential for ADC development. In addition to the primary components of ADCs, we have an extensive track record in enhancing sensitivity, and we can also reduce false negatives through acid dissociation to improve assay reliability.
| Analyte | Our Service |
|---|---|
| ADA (anti-drug antibody) |
|
| Cytokine |
|
| Others |
|
References
- Ministerial Ordinance on Good Laboratory Practice for Nonclinical Safety Studies of Drugs (Ordinance of the Ministry of Health and Welfare No.21)
- Ministerial Ordinance on Good Clinical Practice for Drugs (Ordinance of the Ministry of Health and Welfare No. 28)
- Pharmaceuticals and Medical Devices Agency, "ICH M10: Bioanalytical Method Validation and Study Samples Analysis",
<https://www.pmda.go.jp/int-activities/int-harmony/ich/0090.html >、(accessed 2025.11.28)
Contact Us for Services
