Bioanalysis for Peptide Drugs Using Low-Adsorption LC–MS/MS

Sumika Chemical Analysis Service, Ltd. (SCAS) addresses the analytical challenges associated with peptides and matrix effects by using our optimal sample-preparation techniques. We rapidly develop robust analytical methods to ensure reliable quantification by using advanced systems composed of low-adsorption HPLC and high-sensitivity MS/MS.
Background
Peptide drugs (Peptides) have the advantages of both small molecules (including intracellular target accessibility and ease of chemical synthesis) and large molecules (including high specificity and strong target affinity). With advanced drug-discovery technologies, a diverse array of peptides is under development worldwide.
Quantitative analysis of drug concentrations in biological samples, which often contain various endogenous contaminants requires highly sensitive and selective LC-MS/MS methodologies. In addition, peptides have unique physicochemical structural properties in terms of solubility, stability and adsorption, making their analyses more difficult.
Features
- For peptides that are difficult to analyze because of their high adsorption characteristics, we can minimize their carryover during HPLC analyses and develop precise and accurate quantitative methods by optimizing sample pre-treatment and measurement conditions according to our clients’ target compounds.
- All method validation and actual sample testing are conducted in compliance with relevant regulations, including GLP and the ICH M10 guideline (International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use – M10: Bioanalytical Method Validation and Study Sample Analysis).
Case Studies
The ICH M10 guideline outlines regulatory requirements for both method validation and actual biological sample analyses. It also requires highly-sensitive analytical method development, because peptides often show pharmacological activities at low to moderate concentrations.
1.Response to ICH M10 Requirements
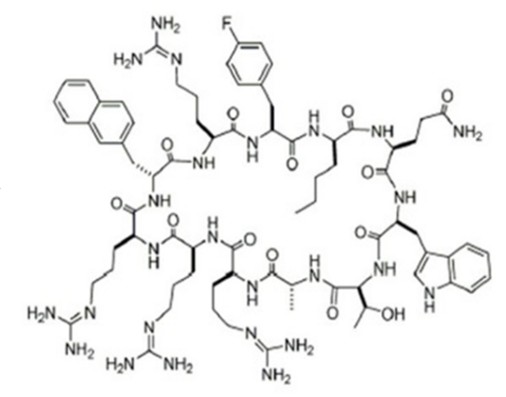
Model Compound: Cyclorasin 9A53
- Cyclic peptide inhibitor of Ras
- Molecular Weight: 1586.849
Cited from Merck
Method Performance and Suitability
- Assay range 1.00~1000 ng/mL
- Linearity Compatibility
- Accuracy Compatibility
- Precision Compatibility
- Selectivity Compatibility
- Carryover Compatibility
Instrumentation
- HPLC: Nexera™ XS inert (Shimadzu Corp.)
- MS: Triple Quad™ 7500 (AB Sciex Pte. Ltd.)
Accuracy and Precision
| Nominal Conc.(ng/mL) | 1 | 2 | 300 | 800 |
|---|---|---|---|---|
| Determined Conc. (ng/mL) | 1.04 | 2.22 | 325 | 887 |
| 1.07 | 2.02 | 330 | 880 | |
| 0.928 | 2.06 | 326 | 854 | |
| 0.908 | 2.21 | 323 | 873 | |
| 1.01 | 2.23 | 323 | 875 | |
| Mean S.D. Precision (%) Accuracy (%) |
0.991 | 2.15 | 325 | 874 |
| 0.07 | 0.1 | 3 | 12 | |
| 7.1 | 4.6 | 0.9 | 1.4 | |
| 99.1 | 107.4 | 108.5 | 109.2 |
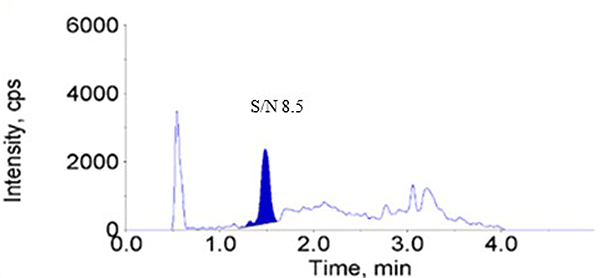
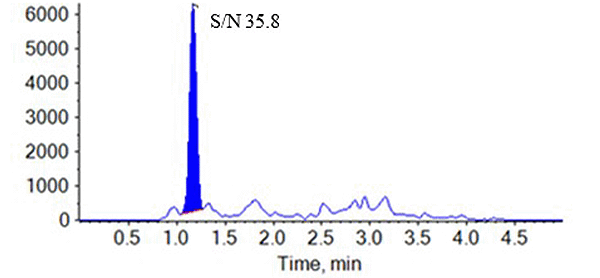
2.Higher Sensitivity
Compared to the previous-generation MS/MS: Triple Quad™ 6500, the latest MS/MS: Triple Quad™ 7500 demonstrates approximately a threefold increase in peak intensity and a fourfold improvement in signal-to-noise ratio (S/N) at the lower limit of quantitation (1.00 ng/mL).
References
- GLP (MHW Ordinance No. 21): "Ministerial Ordinance on Criteria for Conducting Nonclinical Studies on Drug Safety"
- GCP (MHW Ordinance No. 28): "Ministerial Ordinance on Criteria for the Conduct of Clinical Trials on Drugs"
- Pharmaceuticals and Medical Devices Agency: "ICH M10 Bioanalytical Method Validation and Sample Analysis," <000272440.pdf >, (accessed 2025.11.28)
Contact Us for Services
